Fixed Exchange Rate in Pricing of Pharmaceuticals and its Impact
The prices of pharmaceuticals to be launched on the market are determined in accordance with the Decision on the Pricing of Medicinal Products for Human Use (“Decision”) and the Communiqué on the Pricing of Medicinal Products for Human Use (“Communiqué”) dated 29 September 2017, published by the Ministry of Health, which has been authorised to regulate this area.
The Decision foresees a reference pricing system in which the lowest wholesaler price for the relevant product in an EU Member State is used as a reference and is determined to be the maximum possible wholesaler sales price in Türkiye. The reference price is then converted into Turkish lira. However, in order to prevent the impact of exchange rate fluctuations, the aim is to calculate and set a value using an annual fixed exchange rate. This ensures that the exchange rate applied to the reference price from the relevant EU country remains constant for one year, but is significantly lower than the current exchange rate.
According to the Decision, the exchange rate shall be determined as 60% of the average value in Euro from the previous year. The Pricing Evaluation Commission meets within the first 45 days of each year and announces the Turkish lira value for 1 Euro based on the calculation of 60% of the previous year’s average value.
In line with this rule, although the exchange rate used for pricing of pharmaceuticals must be updated annually, updates have been made to the pricing at various times during the year due to the difference between the fixed exchange rate and the current rate at the time, without waiting for the first 45 days of each year.
In this context, a pricing update was implemented in October 2024, and recently this was followed by a further and separate adjustment introduced in December 2025 through an update in the Decision, published in the Official Gazette on 19 December 2025. With this amendment, both the profit margin scales applicable to warehouse and pharmacy sales prices and the Euro exchange rate used in pharmaceutical pricing were revised.
The Turkish lira value of one Euro used in the pricing of medicinal products was increased by 16.9% as of December 2025, resulting in a updated Euro value of TRY 25.3346. The adjustment provides short-term regulatory relief, yet remains an incremental measure rather than a fundamental reform of the underlying pricing policy.
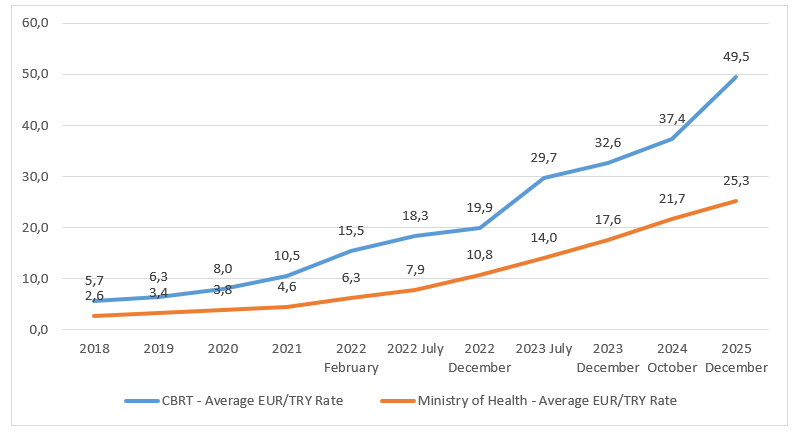
Graph 1. Average Euro/Turkish lira exchange rates according to the Turkish Central Bank (CBRT) and Ministry of Health, TÜRKİYE, 2018-2025
Sources: https://www.tcmb.gov.tr/ and https://www.titck.gov.tr/

